LE SYSTÈME DE CDÉ LE PLUS POLYVALENT POUR VOS ÉTUDES

- Essais cliniques randomisés
- Études cas témoins
- Résultats rapportés électroniquement par le patient (RRP [ePRO])
- Évaluations électronique des résultats cliniques (ÉRC [eCOA])
- Bases de données cliniques administratives
- Études transversales
- Questionnaire en ligne pour patients et médecins
- Études de cohorte
- Registres de patients
- Journaux électroniques
- Études observationnelles
- Analyses rétrospectives des dossiers
- Pharmacovigilance
- Questionnaires en ligne
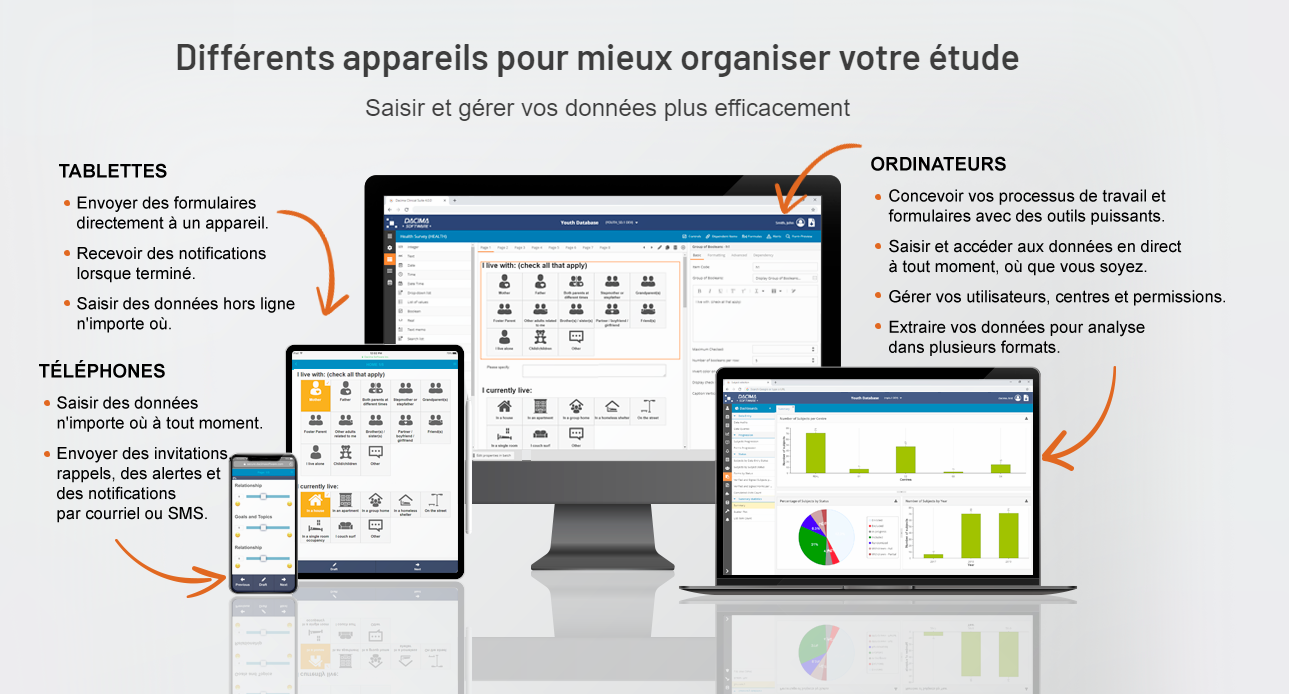
PRINCIPALES FONCTIONNALITÉS
- Interface en ligne configurable
- Formulaire de consentement électronique avec signature
- Capture de données à partir d’images
- Règles de validation et vérification des modifications sophistiquées
- Conforme aux normes E2B pour la pharmacovigilance
- Téléchargement et téléversement de fichiers
- Enchaînements logiques (visible ou activé)
- Filtration par listes déroulantes et listes interrogeables
- Conception de tableaux statiques et dynamiques
- Conception de registres avec regroupements par événements
- Fonctionnalité de journaux électroniques pour patients (par heure ou par jour)
- Programmation de visites et suivi des courriels et des appels de rappel
- Champs sécurisés accessibles aux utilisateurs autorisés seulement
NORMES DE L'INDUSTRIE
PARTENAIRES
CE QUE NOS CLIENTS DISENT DE NOUS
We have been using Dacima for a clinical trial, and needed a full audit trail to comply with FDA guidance for clinical trials. Dacima is a small Canadian company, and it has been a pleasure to work with them on our trial. Their system is most appropriate for longitudinal studies, as it can fully integrate a range of study components longitudinally. Our trial with Dacima is fully integrated into their system, including automated text (SMS) reminders for study visits, electronic consents, electronic CRF, enrollment, randomization procedure, and participant surveys. Dacima’s services were able to fit within an NIH budget for our clinical trial, and they have provided our team at Emory with a fully featured system that meets all our need for the trial. They also have been very responsive in developing functionality particular to our study. I think this is in part because the CEO has a PhD in Epidemiology, so he understands the needs of academic institutions in implementing clinical trials. In sum, I highly recommend Dacima.
Control, ease of use, cost — DACIMA provided a one-stop solution for our clinical data collection: – Control of all aspects of the study / registry from your own offices – Cost is very low compared to other customized solutions – Easy to make edits and duplicate forms – When needed, programmer support is quick and excellent – Wide range of variable types and dependencies – Lots of user control and permission settings – We’ve had good feedback from doctors that the interface and forms looked very professional – Instant compliance with required data management requirements
I’ve been using the software for about 3 years until now, and it helped my team to build up very powerful eCRFs. Our clinical studies became easier to follow. Furthermore, using DACIMA Software helped the investigators to be more efficient at the patient enrolment and monitoring. The alert procedures are very supportive. Moreover, our data manager is closely assisted in forms building.
Dacima offered us a solution tailored to our unique needs, which included limited internet access, inexperienced data entry personnel, a bilingual interface, project-specific data reporting forms and security requirements. Dacima has provided outstanding service. I give Dacima Software my highest recommendation and sincere admiration for its professionalism and dedication to providing work of the highest quality.
I have been a Dacima Software client for more than three years. It has been a great partnership. The Dacima Software people have demonstrated that they are good listeners, action-oriented and client-friendly. I am very pleased with their software and the changes that they have made in response to the changing Study needs.
I have used DACIMA for over 5 years and the platform meets the regulatory requirements needed to support a high standard academic trial. Using DACIMA services is affordable even with an academic budget. The customer service is above expectations.















