
BETTER, FASTER, AND MORE RELIABLE DATA
Dacima’s Electronic Clinical Outcomes Assessment (eCOA) allows clinicians to enter digital data without the need to transcribe clinical data from paper forms and charts. Our solution can be used stand-alone as it is independent of any specific eClinical technologies such as EDC systems, but it can also be integrated into any existing eClinical software landscape.
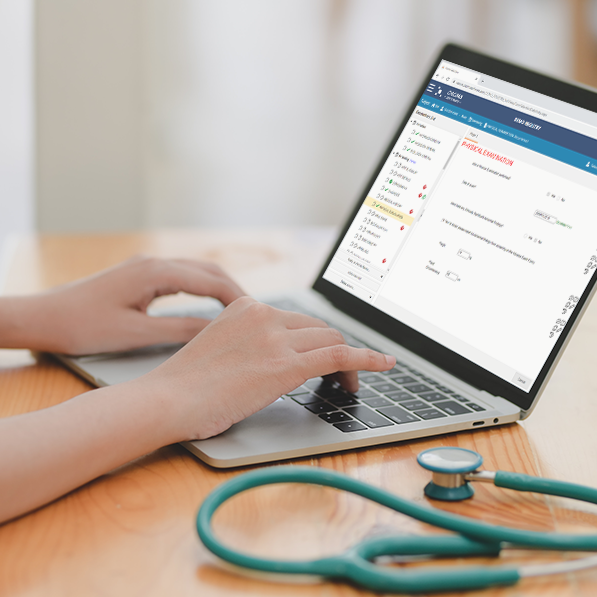
Dacima’s eCOA has been created to collect any reported outcome data in a streamlined, complaint, and efficient way.
Dacima’s web-based eSource software allows for the design and configuration of eCOA forms to meet the specific study protocol requirements through a drag-and-drop designer for rapid setup and implementation. eCOA forms are designed to mimic the familiar paper-pen documents, or through advanced design options the forms can be configured with touchable image icons, edit check and skip logic for visually interactive and intuitive data entry. The web-based software allows clinicians to enter data through their computer, tablet or smart phone via a browser without the need to install apps. The software automatically detects the device and adjusts the display for the type and size of the device.
Dacima’s eCOA software maximizes efficiencies and data quality. The user-friendly technology is ideal for users with any level of research experience and guides proper protocol execution while automatically validating data upon entry.
Advantages:
- Prevents inconsistent or conflicting data
- Eliminates transcription errors
- Meets global regulatory standards

